
I particularly like your find of the Dunnington's method.that method looks pretty clever and I liked learning just a bit about the history of debate that he was invloved with. I like your revised introduction and the new references (though I couldn't download the Nature reference for some reason. This method is the most effective because the frequency can be known quite accurately and the apparatus curvature is fixed, so only the magnetic field needs to be varied and the value of interest will correspond to a sequential quenching of current from the incorrect direction of the beam(4). The quantities measured are, the frequency of an alternating current, the magnetic field at a very specific value that allows for the passage of the beam through a series of slits, and finally the curvature of the apparatus. 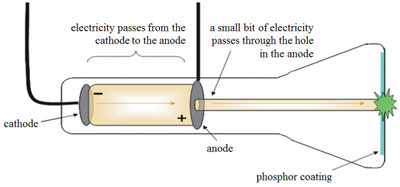
This is similar to the procedure of our apparatus, but differs by the complexity of measurement. Finally, I believe the most effective method of measuring the e/m ratio is from the Dunnington's method. It was discovered in his correspondance with Lorentz, that his method would result in a value for the charge to mass quotient from the magnitude of the effect(5). A more advanced approach was found in the magneto-optical investigation by Zeeman, who discovered that using a Faby-Perot interferometer that the frequency of light is affected by a magnetic field. Another method that closely corresponds to our method is to use a magnetron, which differs in the use of a solenoid, but the data analysis is the same(3). To determine the value of e/m, it is necessary to solve the Lorrentz force equation for the quotient, and resultantly it is necessary to measure the voltage or the accelarating electrodes, current of the Helmholtz coils, and radius of the resulting circle. Our method was to create an electron beam and then manipulate the path of the beam using a magetic field between Helmholtz coils, this produces spirals, that if you rotate the bulb will terminate at the anode(2). The method that Thomson used for his determination of e/m was correcting an electron beam deflected by the electric field by a magnetic one, we avoided this method as the value for the electric field could not be known with much accuracy. Thomson's experiments with cathode rays(1). Thomson used several apparatuses to find useful information regarding this ratio(e/m) and the velocity of the beam, and concluded that information about Q = The quantity of electricity, W = The kinetic energy of the particles, and I = Hρ = The radius of curvature in a uniform magnetic field(H). Our investigation was chiefly to discover the value for the ratio of electric charge to the mass of the particle constituting cathode rays (now identified as electrons after the discovery of the unit of charge of the electron by R. Thus our reasoning for following in his experimental footsteps is to gain further understanding of the scientific method as it relates to the discovery of atomic structure and cathode ray composition. In 1897, Thomson published a paper on the research he had performed to determine the composition of cathode rays, and matter. We also investigated a curious phenomenon which occured in the emmission spectra of the helium excited by our electron beam.įor our laboratory investigation of the nature of cathode rays and the particles composing them we followed the experimental guidelines originally conducted by J.J. We believe our great discrepancy from the accepted value is due to two parts: to the systematic error inherent in the setup and the measurement technique (which was by eye). Our final accepted measured value for the value of e/m(measured) = 4.7(4) * 10^10 C/kg differing from the accepted value of e/m(accepted) = 1.76 * 10 ^11 C/kg. We subsequently measured necessary values and solved the Lorentz force law for the value of e/m. Our procedure created a beam which we manipulated to form a circle by electrodes and Helmholtz coils. We used the experimental layout suggested in Professor Gold's lab manual to do so. With the hindsight knowledge of subsequent studies that the substance in question is composed of a beam of electrons, we conducted our experiment to discover the ratio of the electrical charge to mass for the particle. Thompson to discover certain characteristics for the material referred to as cathode rays. This was an investigation after the method of J.J. I think you did a good job condensing the abstract into your final version.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
